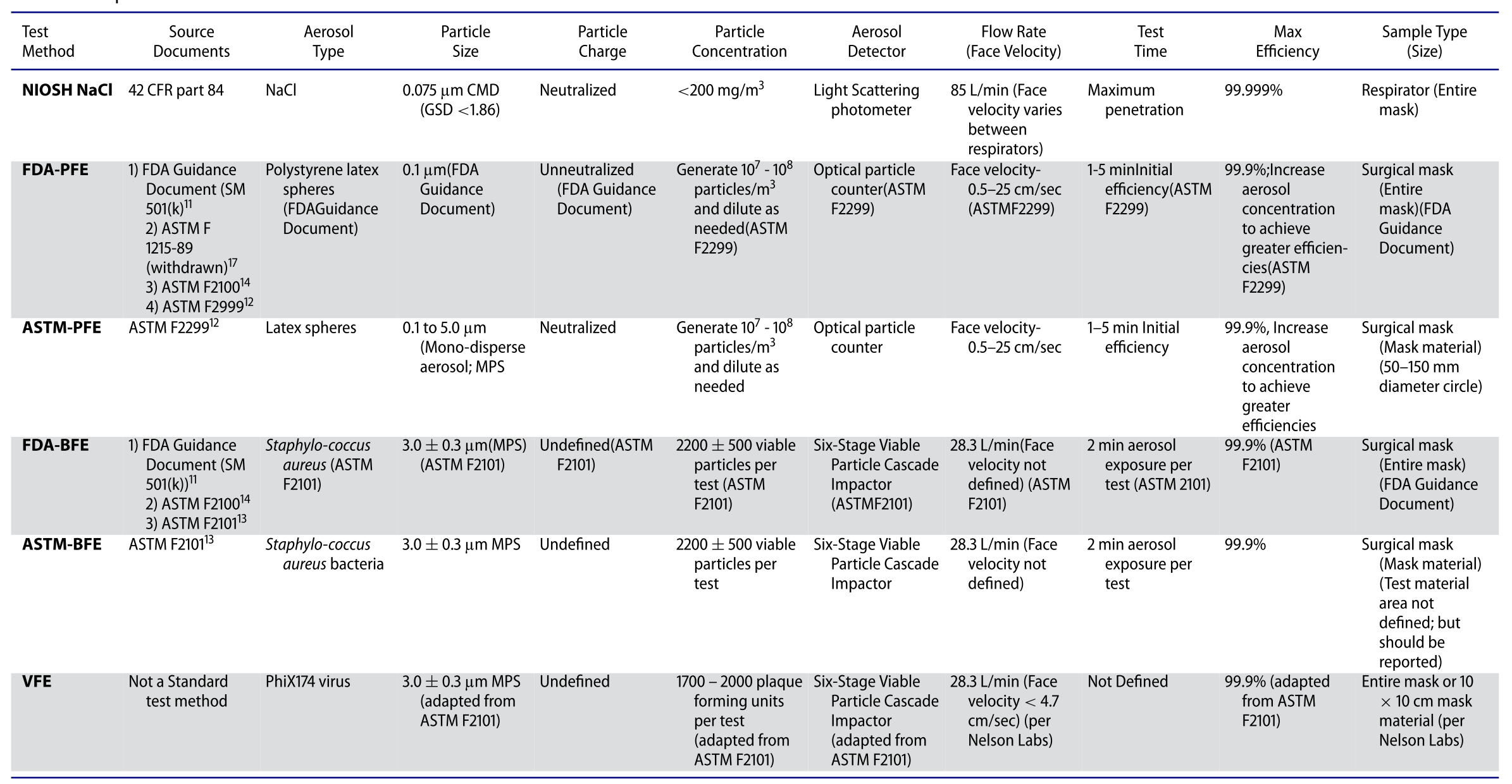
In order to make a proper mask/respirator selection, it is critical to evaluate them with proper test methods for each application. Currently, there are several different test methods to evaluate face masks and respirators, which have different test methods and purposes. Rengasamy et al (2017) compared the mask/respirator filtration test methods as shown in the table below.
NIOSH is the only organization that can certify N95 respirator qualification and their test conditions are considered to be the most stringent by challenging charge neutralized particles with the size close to the most penetrating particle size (MPPS, ~ 50nm for N-type respirators) at higher face velocity to produce conservative filtration efficiency. The face velocity passing through the filter media varies between the respirator types and the face velocity of 3M N95 is calculated to be 9.3 cm/s based on the breathing flow rate (85 lpm) and the respirator surface area (150 cm2). Filter efficiency is measured using an Automated Filter Tester (TSI 8130) with NaCl particles generated from 2% NaCl solution. The particle concentrations in the upstream and the downstream are measured by a light scattering photometer and the filtration efficiency is calculated by comparing the overall concentrations between the two, which gives a single efficiency value for the overall particle size range.
PFE of the different devices was measured using unneutralized 0.1 µm PSL (Polystyrene latex) particles and the test velocity was within the range 1–25 cm/sec as described in the ASTM 2299 standard. The filtration efficiency is calculated by comparing the number concentrations between the upstream and downstream of the test filter.
The Bacterial Filtration Efficiency (BFE) test determines the mask filtration efficiency based on ASTM F2101 standard. A suspension of Staphylococcus aureus is aerosolized using a nebulizer to give a challenge level of 1700–2700 colony-forming units (CFU) per test. These non-neutralized bacterial droplets (Mean particle size 3.0 ± 0.3 µm) are delivered to the filtration media at a constant flow rate of 28.3 lpm (1 CFM). The bacterial droplets are sampled by a six-stage Andersen sampler, which is designed based on the human respiratory tract. The BFE can be calculated by comparing CFUs collected by the six-stage Anderson sampler with/without the test filter media.
The VFE is not recognized as a standard test method but has been adapted by Nelson Laboratories (https://www.nelsonlabs.com) from the ASTM F2101 method. VFE test follows the same procedure as BFE, except the challenge organism is the bacteriophage phiX174 and Escherichia coli bacteria as the host.
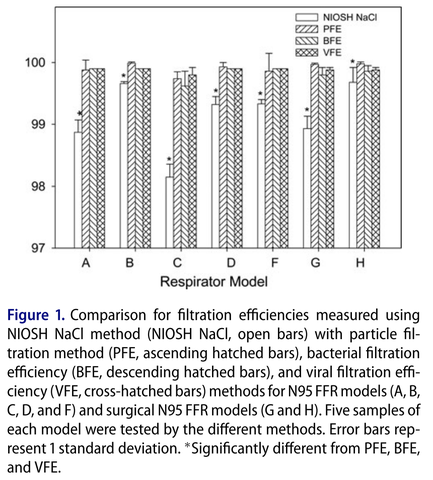
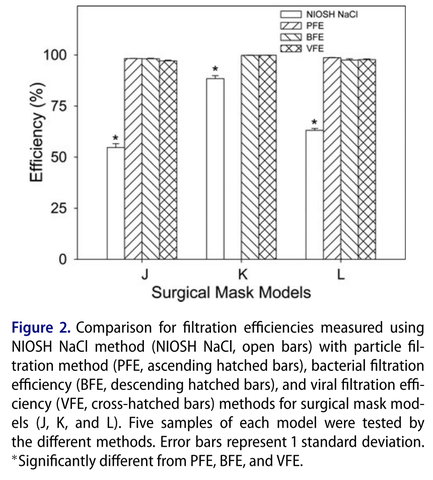
Below are comparison results between the different filtration test methods using various N95 respirators and surgical mask models (Rengasamy et al. 2017)
References
1. Rengasamy S, Shaffer R, Williams B, Smit S. A comparison of facemask and respirator filtration test methods. J Occup Environ Hyg. 2017;14(2):92-103. doi:10.1080/15459624.2016.1225157
2. FDA: “Guidance for Industry and FDA Staff. Surgical Masks - Premarket Notification [510(K)] Submissions; Guidance for Industry and FDA.” Available at http://www. fda.gov/MedicalDevices/DeviceRegulationandGuidance/ GuidanceDocuments/ucm072549.htm.
3. ASTM: ASTMF2299-03, Standard Test Method for Deter- mining the Initial Efficiency of Materials Used in Medical Face Masks to Penetration by Particulates Using Latex Spheres. West Conshohocken, PA:ASTM International, 2003.
4. ASTM: F2101- 01: Standard test method for evaluating the bacterial filtration efficiency (BFE) of medical face mask materials, using a biological aerosol of Staphylococcus aereus. Ann. ASTM Stand. (F2101-01): 1553–1557 (2001).
5. ASTM: F2100: Standard specification for performance of materials used in medical face masks. Ann. Book ASTM Standards, Philadelphia, PA, 2011. pp. 447–449.
6. ASTM:“ASTM F1215 - 89. Test Method for Determining the Initial Efficiency of a Flatsheet Filter Medium in an Air- flow Using Latex Spheres (Withdrawn 1998).” Philadelphia, PA: ASTM International, 2003. (1989).